#BioChemistryOfLife: Composition and Properties of Urine
Nigeria
2
✒️ - By Aan Gray
 The basic composition and properties of urine are as follows:
The basic composition and properties of urine are as follows:
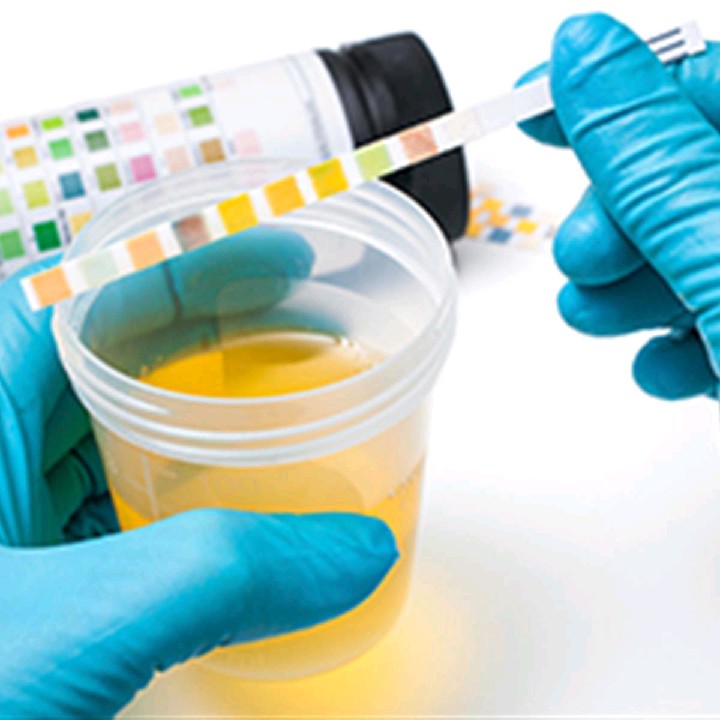
∙ Appearance: Urine varies from almost colorless to deep amber, depending on the body's state of hydration. The yellow color of urine is due to urochrome, a pigment produced by the breakdown of hemoglobin from expired erythrocytes. Pink, green, brown, black, and other colors can result from certain foods, vitamins, drugs, and metabolic diseases Urine is normally clear but turns cloudy upon standing because of bacterial growth. Pus in the urine (pyuria) makes it cloudy and suggests kidney infection. Blood in the urine (hematuria) may be due to a urinary tract infection, trauma, or kidney stones. Cloudiness or blood in a urine specimen sometimes, however, simply indicates contamination with semen or menstrual fluid.
 • Odor: Fresh urine has a distinctive but not necessarily repellent odor. As it stands, however, bacteria multiply, degrade urea to ammonia, and produce the pungent odor typical of stale wet diapers. Asparagus and other foods can impart distinctive aromas to the urine. Diabetes mellitus gives it a sweet, fruity odor of acetone. A mousy odor suggests phenylketonuria (PKU), and a rotten odor may indicate urinary tract infection.
• Odor: Fresh urine has a distinctive but not necessarily repellent odor. As it stands, however, bacteria multiply, degrade urea to ammonia, and produce the pungent odor typical of stale wet diapers. Asparagus and other foods can impart distinctive aromas to the urine. Diabetes mellitus gives it a sweet, fruity odor of acetone. A mousy odor suggests phenylketonuria (PKU), and a rotten odor may indicate urinary tract infection.
∙ Specific gravity: This is a ratio of the density (g/mL) of a substance to the density of distilled water. Distilled water has a specific gravity of 1.000, and urine ranges from 1.001 when it is very dilute to 1.028 when it is very concentrated. Multiplying the last two digits of the specific gravity by a proportionality constant of 2.6 gives an estimate of the grams of solid matter per liter of urine. For example, a specific gravity of 1.025 indicates a solute concentration of 25 × 2.6 = 65g/L.
∙ Osmolarity: Urine can have an osmolarity as low as 50mOsm/L in a very hydrated person or as high as 1,200mOsm/L in a dehydrated person. Compared with the osmolarity of blood (300mOsm/L), then, urine can be either hypotonic or hypertonic.
 ∙ pH: The body constantly generates metabolic acids and gets rid of them by excreting mildly acidic urine, usually with a pH of about 6.0 (but ranging from 4.5 to 8.2).
∙ pH: The body constantly generates metabolic acids and gets rid of them by excreting mildly acidic urine, usually with a pH of about 6.0 (but ranging from 4.5 to 8.2).
∙ Chemical Composition: Urine averages 95% water and 5% solutes by volume. Normally, the most abundant solute is urea, followed by sodium chloride, potassium chloride, and lesser amounts of creatinine, uric acid, phosphates, sulfates, and traces of calcium, magnesium, and sometimes bicarbonate. Urine contains urochrome and a trace of bilirubin from the breakdown of hemoglobin and related products, and urobilin, a brown oxidized derivative of bilirubin. It is abnormal to find glucose, free hemoglobin, albumin, ketones, or bile pigments in the urine; their presence is an important indicator of disease.
 Tags: #ScienceWithGray #ScienceOnBuzz #TheBodyPhysiology #Science
Tags: #ScienceWithGray #ScienceOnBuzz #TheBodyPhysiology #Science

 The basic composition and properties of urine are as follows:
The basic composition and properties of urine are as follows:∙ Appearance: Urine varies from almost colorless to deep amber, depending on the body's state of hydration. The yellow color of urine is due to urochrome, a pigment produced by the breakdown of hemoglobin from expired erythrocytes. Pink, green, brown, black, and other colors can result from certain foods, vitamins, drugs, and metabolic diseases Urine is normally clear but turns cloudy upon standing because of bacterial growth. Pus in the urine (pyuria) makes it cloudy and suggests kidney infection. Blood in the urine (hematuria) may be due to a urinary tract infection, trauma, or kidney stones. Cloudiness or blood in a urine specimen sometimes, however, simply indicates contamination with semen or menstrual fluid.
 • Odor: Fresh urine has a distinctive but not necessarily repellent odor. As it stands, however, bacteria multiply, degrade urea to ammonia, and produce the pungent odor typical of stale wet diapers. Asparagus and other foods can impart distinctive aromas to the urine. Diabetes mellitus gives it a sweet, fruity odor of acetone. A mousy odor suggests phenylketonuria (PKU), and a rotten odor may indicate urinary tract infection.
• Odor: Fresh urine has a distinctive but not necessarily repellent odor. As it stands, however, bacteria multiply, degrade urea to ammonia, and produce the pungent odor typical of stale wet diapers. Asparagus and other foods can impart distinctive aromas to the urine. Diabetes mellitus gives it a sweet, fruity odor of acetone. A mousy odor suggests phenylketonuria (PKU), and a rotten odor may indicate urinary tract infection.∙ Specific gravity: This is a ratio of the density (g/mL) of a substance to the density of distilled water. Distilled water has a specific gravity of 1.000, and urine ranges from 1.001 when it is very dilute to 1.028 when it is very concentrated. Multiplying the last two digits of the specific gravity by a proportionality constant of 2.6 gives an estimate of the grams of solid matter per liter of urine. For example, a specific gravity of 1.025 indicates a solute concentration of 25 × 2.6 = 65g/L.
∙ Osmolarity: Urine can have an osmolarity as low as 50mOsm/L in a very hydrated person or as high as 1,200mOsm/L in a dehydrated person. Compared with the osmolarity of blood (300mOsm/L), then, urine can be either hypotonic or hypertonic.
 ∙ pH: The body constantly generates metabolic acids and gets rid of them by excreting mildly acidic urine, usually with a pH of about 6.0 (but ranging from 4.5 to 8.2).
∙ pH: The body constantly generates metabolic acids and gets rid of them by excreting mildly acidic urine, usually with a pH of about 6.0 (but ranging from 4.5 to 8.2).∙ Chemical Composition: Urine averages 95% water and 5% solutes by volume. Normally, the most abundant solute is urea, followed by sodium chloride, potassium chloride, and lesser amounts of creatinine, uric acid, phosphates, sulfates, and traces of calcium, magnesium, and sometimes bicarbonate. Urine contains urochrome and a trace of bilirubin from the breakdown of hemoglobin and related products, and urobilin, a brown oxidized derivative of bilirubin. It is abnormal to find glucose, free hemoglobin, albumin, ketones, or bile pigments in the urine; their presence is an important indicator of disease.
 Tags: #ScienceWithGray #ScienceOnBuzz #TheBodyPhysiology #Science
Tags: #ScienceWithGray #ScienceOnBuzz #TheBodyPhysiology #Science
Comments (2)
0/500
New Comments(2)
iGrayons
rosemarycw64s:
good to hear
rosemarycw64s
good to hear
Yeah, and thanks.